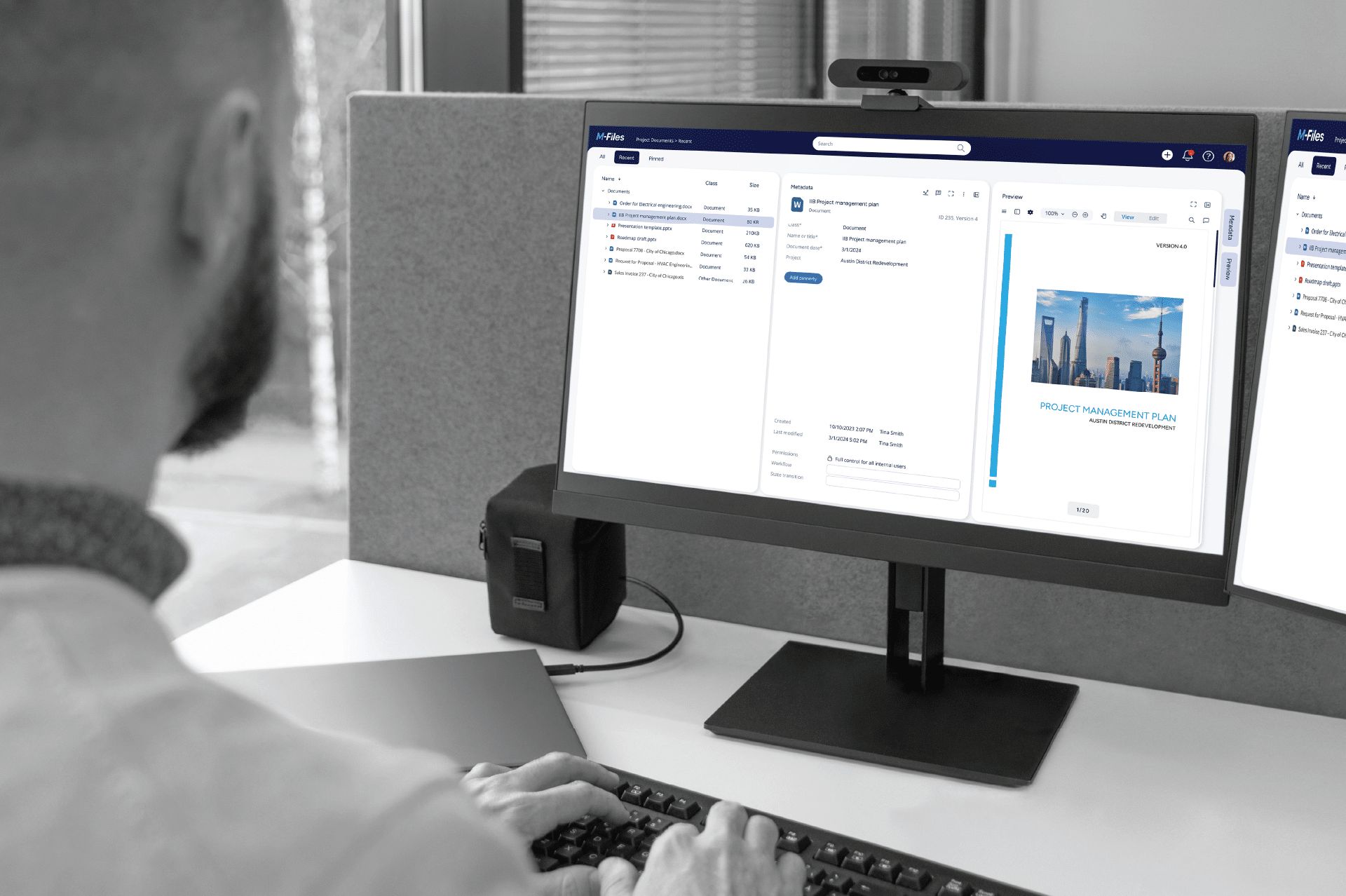
NEWS
Our Insights
M-Files, constantly evolving. Read the latest insights

For many organisations, controlled documents are the backbone of daily operations. Policies, procedures, standard operating procedures (SOPs), work instructions, and forms guide how work is done and how risks are managed. For some organisations, controlled documents underpin important certifications such as ISO 9001, and must be managed very carefully. Yet surprisingly, many businesses still manage these critical documents using shared network drives, spreadsheets, email, or basic collaboration tools. While these approaches may work at a small scale, they quickly become risky as organisations grow, introduce compliance requirements, or operate in regulated environments. This is where a Controlled Document Management System (CDMS) becomes essential. A CDMS is a sophisticated digital platform designed to maintain the integrity of your most critical information. It acts as the "guardian" of your organisation's data, ensuring that critical quality documents are accessible to the right people while being strictly protected from unauthorised access or accidental alteration. A CDMS ensures that documents which govern how work is performed are: Properly created and approved Kept up to date Accessible to the right people Protected from unauthorised changes Fully traceable for audit and compliance purposes In simple terms, a CDMS ensures that only the latest approved version of a document is used , while older (superseded) versions are retained securely for reference and audit history. Superseded versions are typically available only to administrators. Controlled document management systems are commonly used by organisations that need to comply with standards such as ISO 9001 or other quality, safety, and regulatory frameworks.

For any organisation committed to excellence, the quality management audit is a critical test of process integrity and discipline. While the thought of an audit can be daunting, its success often hinges on one foundational pillar: rigorous document control . This isn't just about having files in folders; it's about demonstrating a complete, traceable, and verifiable history of every procedure, record, and change. Without this control, an organisation walks into an audit unprepared, risking findings that can lead to severe consequences. Failing an audit is more than just a mark on a report. It can result in the loss of crucial certifications (like ISO 9001), operational shutdowns, damaged customer trust, and a significant hit to both your reputation and bottom line. In this article we take a deep-dive into the popularity and risks of SharePoint as a platform for managing version-controlled documents, such as quality documents. We contrast those risks with the capabilities of M-Files as a document management platform designed specifically with compliance and audits in mind.

We hear similar frustrations time and time again: “ managing access to documents in our SharePoint is a nightmare! ”. People come to us all the time looking for answers because SharePoint admins are tearing their hair out trying to help team members with accessing documents they need to do their jobs effectively. The crux of the problem is that SharePoint is essentially a folder-based document management platform. As we have written about previously , be it in SharePoint or a local network drive, folders are rigid file management structures that cause headaches, regardless of how many times you tinker with your organisation’s folder structure. In this article, we use a case study to contrast document access permission approaches between SharePoint and M-Files. As you will see, the metadata-driven approach adopted by M-Files provides a supremely flexible framework that solves common SharePoint access problems, saving your admins time and your organisation money.

Effective document management is crucial for successful project management, ensuring smooth operations and clear communication. Project document management involves organising, storing, and tracking all project-related documents. A good project document management solution makes essential information easily accessible to relevant personnel and external stakeholders to prevent costly delays. Proper project document management enhances collaboration, improves efficiency, and ensures compliance with regulatory requirements. Managing the sheer volume of documents in a project can be challenging with traditional methods. Information can be hard to find, leading to wasted time and duplicated efforts. Version control issues are rampant when documents aren't found or versions are tracked manually through ambiguous file naming, increasing the risk of team members making decisions based on outdated information. External collaboration often requires separate systems, adding more work and compounding issues with duplication, version control, and visibility.

Aged care and community care providers in Australia play a vital role supporting vulnerable individuals. However, the sector faces unique and complex challenges when it comes to managing documents and information. Dealing with sensitive personal health data and adhering to strict compliance requirements can often lead to administrative burdens that detract from the core mission of providing quality care. In this post we explore some of the key information management challenges faced by the aged care and community care (ACCC) sector and show how M-Files can help ACCC providers enhance regulatory compliance and streamline their internal operations.

Registered Training Organisations (RTOs) in Australia play a vital role in skilling the nation's workforce. However, they must navigate a complex landscape of regulatory requirements, increasing data volumes, and the constant need to maintain accurate and accessible information. For many RTOs, this often translates into a struggle with inefficient document management practices, hindering productivity and potentially impacting compliance. In this post we will explore some of the key information challenges faced by Australian RTOs and how an information management solution like M-Files can provide a powerful pathway to streamlined operations and enhanced success.

For Australia’s burgeoning construction and manufacturing sectors, efficient management of information is no longer a luxury – it is a fundamental requirement for success. From complex project blueprints and safety regulations to intricate manufacturing specifications and quality control records, these industries generate vast quantities of critical documentation. Without a robust system to organise, secure, and access this information, construction and manufacturing businesses can face significant hurdles, impacting productivity, compliance, and ultimately, profitability. Australian construction and manufacturing businesses operate within strict regulatory frameworks. Construction projects in Australia are subject to a myriad of regulations, including building codes, environmental protection laws, and workplace health and safety acts. For instance, ensuring all permits, contracts, safety reports, and quality certifications meet the required standards is crucial to avoid legal penalties and project delays. Similarly, Australian manufacturers often adhere to industry-specific standards, quality control regulations like ISO 9001, and workplace safety legislation. Managing the documentation required to demonstrate compliance with these evolving regulations can be a significant undertaking. This post discusses five common document management challenges faced by Australian construction and manufacturing businesses. We also discuss how M-Files , a powerful intelligent information management platform, helps address these common challenges:

Knowledge workers often grapple with the burden of repetitive administrative duties. A prime example is the generation of standard documents such as contracts, reports, and proposals. These tasks frequently involve manual data entry, which wastes valuable time and can lead to data entry errors. Fortunately, M-Files offers a robust solution with its intelligent document templates that automatically populate placeholders with relevant metadata, significantly speeding up document creation.

Knowledge workers often waste significant time searching for necessary documents. Traditional folder-based systems can become disorganised, leading to lost productivity, duplicated efforts, and user frustration. Research shows that many employees waste hours each week struggling to find documents and may even recreate them due to ineffective systems. It is essential for knowledge workers to focus on their core responsibilities rather than being bogged down by inefficient document retrieval processes. Fortunately, modern information management platforms such as M-Files offer intelligent features that revolutionise information management , significantly reducing search times and improving overall efficiency. Here's how M-Files empowers users to locate documents quickly and accurately:
The sheer volume of email can be a significant drain on productivity. Hours are often consumed by drafting, sending, sorting, and searching through a barrage of messages. This constant engagement with email diverts attention from core responsibilities and more strategic work. Furthermore, utilising email for the exchange of sensitive information and critical documents introduces considerable security risks with unencrypted emails being susceptible to interception. Vital documents can become buried within lengthy email threads, making their retrieval a cumbersome and often frustrating task. This blog post will delve into how M-Files, a sophisticated information management platform , presents a more efficient and secure approach to managing many tasks traditionally handled through email. By capitalising on specific functionalities, organisations can lessen their reliance on email, recover valuable time, and bolster their data security. Here are five M-Files features designed to help your team reduce email overload:

Email has become the ‘go-to’ tool for communication within organisations. Email often serves not only as a means of immediate correspondence but as a pseudo repository for critical business information, even a makeshift file storage system. While email is no doubt convenient, reliance on email for managing business information presents significant challenges that can impede productivity, jeopardise security, and negatively affect an organisation’s overall efficiency. In this post we explore the challenges and risks of over-reliance on email for managing critical business information. We also discuss how M-Files offers a comprehensive solution for addressing challenges and risks associated with email.

Knowledge workers spend a significant amount of time searching for documents, impacting productivity and costing organisations money. Research indicates professionals spend an average of 18 minutes locating a single document . This wasted time adds up across the course of a month or year, with estimates suggesting time wasted searching for documents costs organisations nearly US$20,000 per knowledge worker annually . For many organisations, inefficient, folder-based document repositories are the status quo, and the impact of these inefficiencies is rarely questioned. M-Files offers a solution to this problem through a new paradigm in information management: metadata. Metadata, or “data about data”, allows for intuitive classification and management of documents based on what they are, rather than where they are stored. This approach delivers significant time savings in document retrieval.

Effectively managing document versioning presents a significant challenge for organisations. Poor version control can lead to confusion, errors, and inconsistencies that increase costs and risks. Implementing robust version control practices is, therefore, essential for maintaining organisation, accuracy, and consistency across teams. Without it, organisations risk confusion, wasted time, and potential compliance issues. The good news is that knowledge workers and their organisations can avoid version control problems by implementing a robust information management system. These platforms centralise document storage, automate version tracking, and control access, ensuring that everyone works with the most up-to-date and accurate information. In this post we’ll delve more into the consequences of poor version control and how market-leading information platform M-Files can help eliminate these problems.
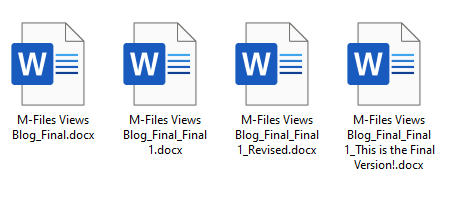
Imagine a world where your team can instantly find any document they need, no matter where the file is stored. That's the promise of a modern Document Management System (DMS) . A DMS is a software solution that helps businesses organise, secure, and manage their documents. What’s more, a modern DMS is often packed with other features such as document workflow management, to drive productivity up. Many traditional DMS solutions rely on a folder-based structure , mimicking physical filing cabinets. So many of us are brought up using folders to store our documents using the ‘File > Save As… > Pick a folder > Give the document a name > Click Save’ mantra. It’s the status quo. But the reality is that the status quo is holding your team back and wasting their time.

Controlling access to sensitive information is paramount for organisations who rely on knowledge for value creation and competitive advantage. Data breaches are increasingly common, and regulatory requirements are becoming more stringent. A robust document management system (DMS) is crucial for protecting your organisation’s data and meeting regulatory obligations. A DMS provides a centralised repository where documents can be stored, organised, and accessed by authorised users. Choosing the right DMS is essential, and understanding how access permissions work within these systems is key. In this post we will introduce M-Files as a market-leading DMS platform and discuss how access permissions can be managed within M-Files. We will also compare and contrast approaches to access permissions between M-Files and the other well-known DMS platform, Microsoft SharePoint.

Maintaining control over critical business documentation is more important than ever. For organisations striving for quality and consistency, controlled documents are essential. But what exactly are controlled documents, and how can M-Files help streamline their management, especially when aiming for ISO 9001 certification? Let's explore.

One of the most powerful features of M-Files is its robust workflow automation capability. In this post, we'll explore what M-Files workflows do and highlight their automation potential. Moreover, we recently wrote about how M-Files can turbocharge your organisation’s SharePoint . In this post we’ll also demonstrate why the M-Files workflow engine surpasses SharePoint workflows.